Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. But did you know that PARP-1, a type of protein, constantly repairs our damaged DNA?
The PARP-1 is a protein found in our cells that constantly repairs damaged DNA. However, it cannot repair the DNA in neurons while we are awake. As a result, it builds up until we fall asleep. According to new research, the accumulation of PARP-1 causes us to sleep
What is the PARP-1 Protein, and Where Do Most of Its Activity Occur?
PARP-1, also known as NAD+ ADP-ribosyltransferase 1 or poly ADP-ribose synthase 1, is an enzyme encoded by the PARP1 gene in humans. It is the most abundant of the PARP family of enzymes, accounting for 90% of the NAD+ utilized by the family.
The PARP-1 is found in place of histone H1 in active genes. It is found in the cell nucleus and can detect and respond to metabolic and chemical on a cellular level immediately. (Source: Royal Society of Chemistry)
How Does the PARP-1 Protein Improve DNA Repair in Neurons by Promoting Sleep?
The characteristics of sleep drivers and the mechanisms by which sleep relieves cellular homeostasis pressure are unknown. DNA damage levels in flies, zebrafish, mice, and humans increase during wakefulness and decrease during sleep. We show that 6 hours of consolidated sleep is enough to reduce DNA damage in the zebrafish dorsal pallium.
Sleep and DNA repair were triggered by the induction of DNA damage by neuronal activity and mutagens. The DNA damage response (DDR) proteins Rad52 and Ku80 increased during sleep, and chromosome dynamics increased Rad52 activity.
Following sleep deprivation, the activity of the DDR initiator PARP-1 increased. PARP-1 promoted sleep in both larva zebrafish and adult mice. Its inhibition reduced sleep-dependent chromosome dynamics and repair.
These findings show that DNA damage is a homeostatic driver of sleep and that PARP-1 pathways can detect this cellular pressure and facilitate sleep and repair activity. (Source: X-MOL)
How Does the PARP-1 DNA Detect Damage and Repair Cells?
By forming the posttranslational modification poly ADP-ribose, PARP-1 affects gene transcription, cell death signaling, and DNA repair ADP-ribose. PARP-1 is rapidly associated with DNA damage during the physiological response to genotoxic stress, resulting in a powerful stimulation of poly ADP-ribose synthesis over a low basal level of PARP-1 activity.
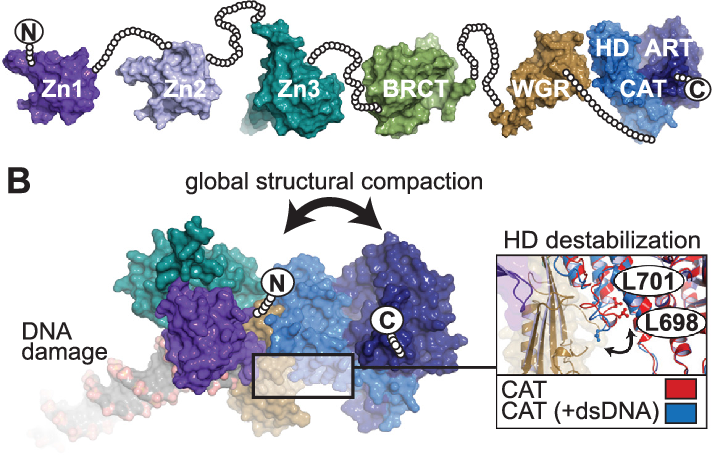
Understanding PARP-1’s biological function is dependent on DNA damage. Still, structural insights into the mechanisms behind this manner of regulation have remained elusive, partly because of PARP-1’s extremely modular six-domain architecture. PARP-1 uses unique zinc fingers to detect DNA breaks through sequence-independent interactions with exposed nucleotide bases, a typical hallmark of damaged and aberrant DNA structures, according to recent structural investigations.
Based on a crystal structure of the critical domains of PARP-1 in association with a DNA strand break, the method of connecting DNA damage detection to enhanced poly ADP-ribose production has been revealed. PARP-1’s many domains collide with damaged DNA, generating a network of interdomain interactions that cause destabilizing changes in the catalytic domain, resulting in increased poly ADP-ribose synthesis. (Source: National Library of Medicine)